We use cookies to help improve our website and personalise your experience. To find out more about how to manage these cookies, please see our cookie policy.

We are very pleased to report that as a result of our recycling and waste management programme, implemented together with Recycling Lives on two recent construction projects, we supported the delivery of more than 700 meals to vulnerable communities across Lancashire and Cumbria...READ MORE

We are pleased to announce that we have been awarded a contract for the design and construction of a new Quality Assurance Laboratory for a pharmaceutical blue chip client. Employed as principal designer and principal contractor, our dedicated team will deliver this project on a full turnkey basis, including civil and structural, architectural, HVAC and electrical services...READ MORE

We are very pleased to announce that we have completed a viral vector suite facility for Cobra Biologics (“Cobra”); a biologics specialist at the forefront of COVID-19 vaccine scale-up and manufacturing. Located within Keele University campus, Cobra has invested in upgrading part of its existing development and production facility with 3 viral vector suites to enable the company to expand its clinical and commercial viral vector services as a Contract Development and Manufacturing Organisation (CDMO). The time-critical project is a pivotal element of the company’s commercial strategy and BES has delivered the project despite the social distancing and supply chain challenges of the COVID-19 pandemic.

BES has successfully completed the first phase of a project to design and construct a new pharmacy and aseptic suite at Weston Park Hospital in Sheffield. The project includes clinical trial and ATMP (Advanced Therapy Medicinal Products) facilities that will enable the hospital to provide gene therapy treatments on site. The project is being delivered in two coordinated phases to allow the existing pharmacy to remain operational while the new one is delivered, before work begins on the aseptic suite...READ MORE

BES has recently completed the modernisation of the new Protein Science Laboratories for AstraZeneca, within their facility at Granta Park in Cambridge. As part of AstraZeneca’s UK strategic goal, scientists have moved from the existing Alderley Park research and development laboratories in Cheshire to this new facility, which has been put to immediate use in supporting efforts to develop a vaccine for COVID-19. Working as principal designer and principal contractor, our multidisciplinary team reconfigured the existing space to accommodate the new Protein Science laboratories and coldroom. READ MORE...
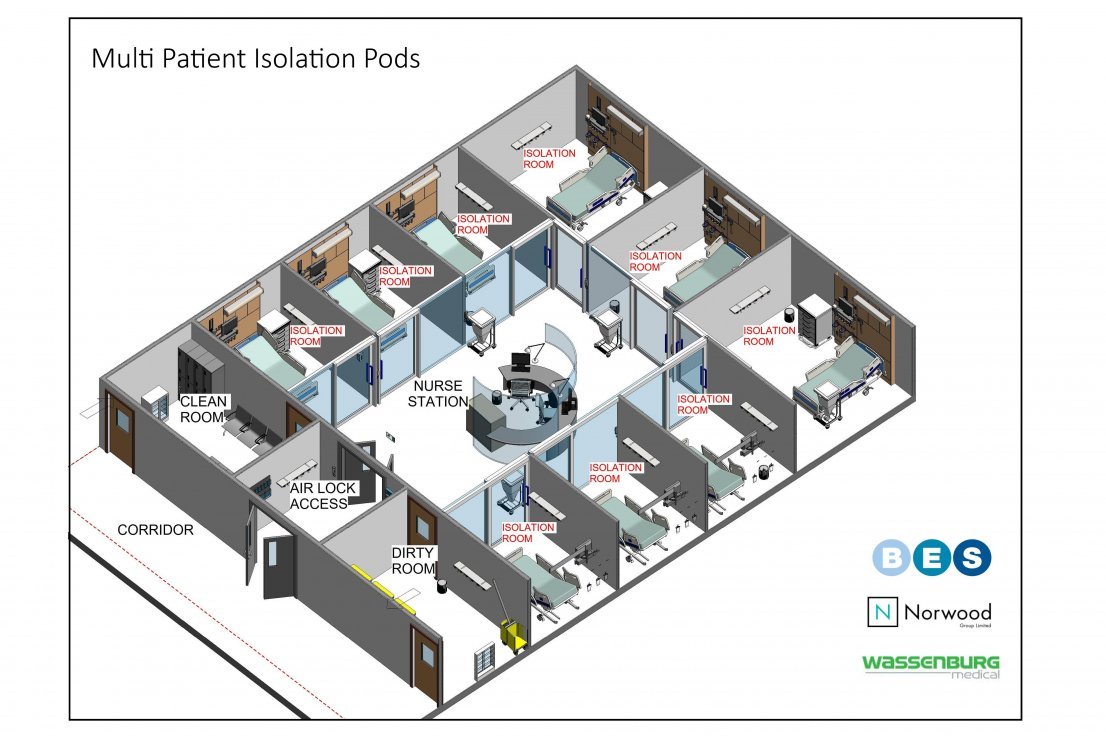
BES and Norwood have teamed up with Wassenburg Medical to combine our manufacturing experience and industry knowledge in order to produce a range of bespoke isolation pods. Developed utilising Norwood’s specialist partitioning systems, the pods can help to battle the spread of coronavirus across the UK. These fully demountable units can be installed into any fallow area within a hospital or can be provided as an external facility to form a temporary isolation ward or a testing station. The flexibility of the construction allows each system to be pre-engineered and tailored to suit any requirements, incorporating solid or glazed panels. We can also provide a range of integrated services which can be concealed and housed within the pod’s structure, delivering a complete solution. All the products are manufactured off site at Norwood’ production facility, thus the isolation pods can be supplied within days, with minimal disruption to ongoing operations.

We are delighted to announce that we have completed a complex £50 million project to upgrade and future-fit the warehousing, packing and logistics facilities at AstraZeneca’s Macclesfield site. Working collaboratively with the pharmaceutical giant as an integral part of the company’s user, quality assurance and project management team, BES was principal designer and principal contractor on the three-phase programme. The project involved full design, construction and commissioning of a new high bay warehouse, a low bay warehouse refurbishment and a technically-demanding Global Packing Centre.

We are pleased to report that we have completed an £8.1 million design and build project to deliver Compound Semiconductor Applications (CSA) Catapult’s facility in the world’s first compound semiconductor cluster in south Wales. The world-leading Catapult Innovation Centre will drive advances in compound semiconductor technology and applications, enabling the UK to spearhead future technologies, including autonomous vehicles, 5G mobile networks, renewable energy and the Internet of Things (IoT). READ MORE...

If you are interested in the design and construction of diagnostic treatment centres and imaging facilities, come and join us at the next HCN breakfast, which will take place on Friday 14 Februrary in RICS, London. Our Southern Regional Director, Gavin Statham and Joshua Bannister from Norwood will be there to share their specialist knowledge and experience of delivering complex sophisticated facilities.

As we look forward to an exciting New Year, we have reviewed last years’ key accomplishments which are summarised in our infographic.